Changes to cancer treatment to have devastating effect on patients
4 Feb 2015 | Stephanie Prior
Table of Contents
The Government’s Cancer Drugs Fund (‘the Fund) announced that 21 out of 84 cancer treatments would be scrapped to save £80 million. The savings are to be then used to introduce new cancer treatment programmes. While existing patients will continue to receive treatment with the 21 ‘axed’ drugs.
It is reported that the changes introduced by the Fund will affect as many as 8,000 cancer patients a year. This will have a devastating effect on patient treatment and will take away their chances of successful treatment. It is further reported that the changes will especially affect patients in more advanced stages of cancer.
Life-prolonging treatments like Jevtana (for prostate cancer, giving at least extra three months of life, costing around £22,000 for a one-off treatment), Zaltrap (for bowel cancer, giving at least extra six weeks of life, costing around £6,000), Halaven (for breast cancer, giving up to extra five months of life, costing around £10,000 for six months treatment), Yondelis (for ovarian cancer, giving up to extra six months of life, costing £57,000 for 18 months treatment) would not be prescribed to newly diagnosed cancer patients from March 2015.
While the costs of the above-named treatment seem at first blush to be very high in comparison with the relatively small benefit in terms of time, the personal element of the extra time afforded to cancer sufferers and their families is priceless.
Another change introduced in 2015 relates to the method of referral for cancer diagnosis. A new (trial) NHS scheme will allow patients to self-refer for a test if they believe they have cancer symptoms. The aim of the scheme is to cut out the delays associated with GP referrals for tests. While it might be the case that it will be possible to eliminate the delay in being tested, this is likely to be only temporary. Self-referral might lead to over-reliance on the service (and abuse), leading to long waiting lists for a test as well as a significant increase in costs.
Furthermore, Cancer Research UK, Macmillan Cancer Support, Public Health England and the Royal College of General Practitioners will unite their powers and work on introducing significant changes to the cancer services in the UK, focusing on prevention, diagnosis, treatment and mental support. This project came about as the UK cancer survival rates are amongst the worst in Europe (especially regarding lung cancer).
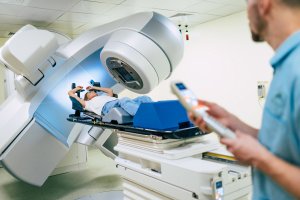
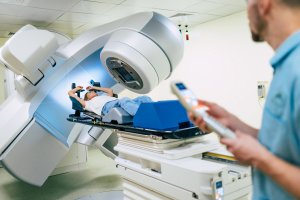
Last but not least, the Department of Health has indicated that the proton beam treatment will be available in hospitals in London and Manchester from April 2018. This is good news.
Proton beam therapy is a type of radiotherapy that uses beams of protons (as opposed to high energy X-rays as in cases of traditional radiotherapy) for killing cancerous cells without damaging the surrounding tissue and organs or causing side effects. This is because it is possible to operate protons more precisely by aiming the therapy at cancerous cells rather than using X-rays.
Once the beams of protons meet the cancerous cells, the proton beam stops at the cancerous cells without moving onto and attacking healthy tissue. Because of this special feature, proton beam therapy is mainly used for the treatment of brain cancer, cancer in the central nervous system or eye cancer.
Proton beam therapy became famous in the UK because of one little boy called Ashya King. Ashya had brain cancer (medulloblastoma) and when his parents did not see a future within the NHS in respect of his cancer treatment, they took him to Prague for proton beam treatment. Such treatment was not available in the UK. It was reported that Ashya reacted well to the treatment. However, his family did not return to the UK because of fears of prosecution (for removing Ashya from the NHS hospital without the treating doctor’s permission).
There is only one clinic in the UK offering proton beam therapy, the Douglas Cyclotron, for eye cancer only. It is also possible to apply for NHS funding for the treatment abroad, although the process might be long and complicated, and the patient may even die before any such referral. However, once an application is made and supported by a consultant, there are good chances it would be further approved for funding. Statistically, since 2008 around 75% of all referrals were approved for funding.
The introduction of proton beam therapy in 2018 would be a great step forward for cancer treatment in the UK. However, the burning question is why it has taken so long and why the UK is so far behind other European countries like the Czech Republic, where proton beam therapy has been available since 1992.
If you think you have suffered substandard medical treatment or significant delay in diagnosis hospital or by a GP or other medical professional, contact Osbornes Medical Negligence department for a free and confidential conversation at 020 7485 8811.
Share this article
Contact
Related InsightsVIEW ALL
- 21.9.2023
Large Compensation for Delayed Laryngeal Cancer Diagnosis
Actress receives financial award after life-changing missed cancer diagnosis. Jodi Newton acted for a client who was belatedly diagnosed with...
Read more - 21.9.2023
Delayed cervical cancer diagnosis claim
Jodi Newton acted for a woman who died following a delayed cervical cancer diagnosis. The deceased could have been diagnosed...
Read more - 13.9.2023
Delayed skin cancer diagnosis
We are pleased to have successfully settled a client’s claim for a delayed cancer diagnosis – Basal Cell Carcinoma (“BCC”)....
Read more - 13.1.2023
Delayed diagnosis of appendiceal cancer
The medical negligence team at Osbornes Law has recently settled a case involving a patient who passed away following a...
Read more - 9.11.2022
Breast cancer screening mammograms and negligence
Breast cancer screening has improved significantly in the UK due to research bettering the understanding of this terrible disease, which...
Read more - 11.4.2022
Late Cancer Diagnosis Crisis Within NHS
The Health and Social Care Committee recently released a 52-page report on cancer services, expanding on the crisis in England (...
Read more - 23.2.2022
Record high waiting lists put cancer patient lives...
In early January 2022 there were nearly six million people in England waiting for routine operations and treatment: a record high....
Read more - 31.1.2022
Melanoma misdiagnosis negligence claim
Jodi Newton is acting for a client with a melanoma on her foot, which the dermatologist diagnosed as benign. Concerned...
Read more - 12.11.2021
Preventing lung cancer delayed diagnosis & misdiagnosis
Lung cancer is one of the most common and serious cancers – sadly, it is often incurable. In common with other...
Read more - 21.9.2020
Delayed Glioma Diagnosis
Our delayed cancer diagnosis solicitors are acting for a family who experienced delays in cancer diagnosis, in that a male...
Read more - 2.9.2020
Cervical cancer misdiagnosis
Cervical Cancer Misdiagnosis Dating Back Four Years Jodi Newton, clinical negligence solicitor Osbornes Law, recently settled a claim for the...
Read more - 6.9.2019
Delay in Cancer Diagnosis and Chances of Survival
According to Macmillan Cancer Support, there are an estimated 2.5 million people living with cancer in the UK. It is predicted...
Read more - 13.8.2019
Claims Against Ian Paterson for Breast Surgery Errors
Former Consultant surgeon Ian Patterson has been found guilty of 17 intentional wounding counts. In May this year he was sentenced...
Read more - 7.5.2018
The NHS, Computers and Breast Cancer
According to reporting from the BBC, as a consequence of a computer error, nearly half a million women in England...
Read more - 12.12.2017
New treatment for Prostate Cancer Patients
The Royal Marsden Hospital and Guys Hospital, two leading London centres of excellence have just joined an exciting new medical...
Read more - 22.4.2015
Bowel Cancer need not be a killer
Early diagnosis of bowel cancer is key. Cancer is a word that attracts stigma. Many people believe that if you...
Read more - 4.2.2015
Changes to cancer treatment to have devastating effect...
The Government’s Cancer Drugs Fund (‘the Fund) announced that 21 out of 84 cancer treatments would be scrapped to save £80 million....
Read more